Computational diagnosis of retinal diseases using deep learning techniques on
optical coherence tomography
In the developed world, diabetic retinopathy is the leading cause of blindness among the working age population. Of an estimated 285 million people with diabetes worldwide, nearly 10% are afflicted with a vision-threatening disease, diabetic macular edema (DME) being the leading etiology. The diagnosis and management of DME is based on optical coherence tomography (OCT). This imaging modality, based on optical reflectivity, is used to obtain cross-section and three-dimensional high-resolution images of the macula. Currently, OCT-derived measures are the standard of care in diagnosing DME and monitoring of therapeutic effects, with thickness measurements of the macula serving as the primary clinical measure.
However, the morphology of DME extends to additional characteristics such as the presence of intraretinal fluid and retinal layer disorganization. It is hypothesized that identification of such features on OCT would enable more precise quantification of DME, and will enhance management decisions. Yet, the analysis of OCT and identification of pathologies is complex and requires highly trained retina experts. Manual interpretations are extremely time consuming, with variable repeatability and interobserver agreement.
Computer-Aided Diagnosis (CAD) has a promising potential for facilitating interpretation of medical imaging, with rising global interest. The field of computer vision has been revolutionized by deep learning methods, with outstanding results in image classification, detection and segmentation. The learning methods have a strong potential to improve diagnosis based on 3D clinical data. To date, no such approach has matured into implementation in the clinical practice of retinal medicine.
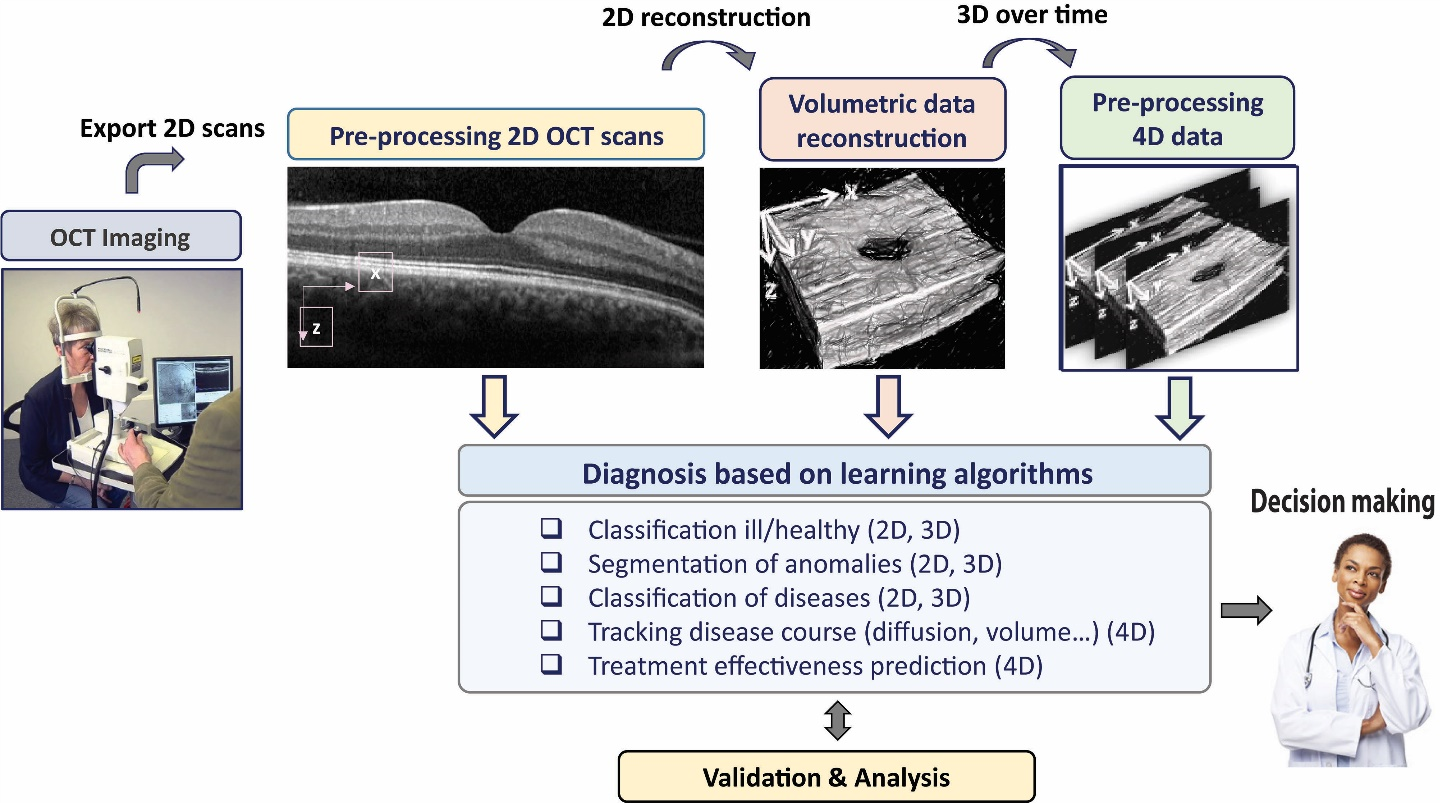
The objective of the proposed research is to promote the clinical care of DME by providing computational-based quantitative and qualitative analysis of OCT. Our approach will apply novel deep-learning methods on OCT data, including volumetric and longitudinal data. A new deep learning method will be developed on 2D OCT images, and will thereafter be extended to OCT volumetric data. Subsequently, a unique model for analysing volumetric OCT data longitudinally (4D data) will be examined to track the disease course and to assess for possible prognostic factors. All results will be analysed and validated by medical experts. We expect that deep learning-based CAD will not only augment the diagnostic tools and enhance improved clinical outcomes in DME, it may also provide a conceptual approach towards similar challenges in additional retinal sight-threatening conditions.
DME analysis from OCT scans – Solution approach